Category: Drug Side Effects

Andexxa Recall Class Action Lawsuit
AstraZeneca has recalled Andexxa after finding the drug increased the risk of serious blood clots, stroke, heart attack, and death. If you or a loved one received Andexxa and experienced serious clotting complications or death, you may be eligible for...

Ocaliva Class Action Lawsuit
Intercept Pharmaceuticals has withdrawn Ocaliva (obeticholic acid) from the U.S. market following FDA warnings of serious liver injury in patients without cirrhosis, affecting thousands who relied on the drug for primary biliary cholangitis (PBC) treatment. This action, amid reports of...
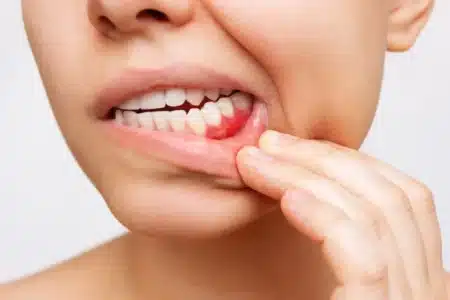
Suboxone Tooth Decay Class Action Lawsuit
Recent studies have found that long-term use of the Opioid Use Disorder (OUD) drug Suboxone can increase the risk of tooth decay and other serious dental side effects.

Wegovy Stomach Paralysis Class Action Lawsuit
Our law firm is reviewing potential lawsuits on behalf of people who developed stomach paralysis or "gastroparesis" after taking the weight-loss drug Wegovy (semaglutide), because the manufacturer failed to adequately warn about the risk of this dangerous gastrointestinal side effect.
Rybelsus Stomach Paralysis Class Action Lawsuit
Recent lawsuits allege that Rybelsus (semaglutide), a type 2 diabetes drug that is commonly prescribed "off-label" for weight-loss in obese patients, can cause stomach paralysis, a disorder that stops or slows the movement of food from the stomach to the...

Mounjaro Stomach Paralysis Class Action Lawsuit
Eli Lilly's new type 2 diabetes drug Mounjaro (tirzepatide), which is also commonly prescribed "off-label" for weight-loss, has recently been named in a lawsuit alleging that the medication increases the risk of a severe gastrointestinal disorder called stomach paralysis, also...
Ozempic Stomach Paralysis Class Action Lawsuit
Lawsuits are now being filed against the makers of Ozempic (semaglutide), a type 2 diabetes drug which is also prescribed "off-label" for weight loss, that has been linked to a severe gastrointestinal problem called stomach paralysis, which is also known...

Weight Loss Drug Stomach Paralysis Class Action Lawsuit
Lawsuits are now being filed against the manufacturers of a line of widely-prescribed type 2 diabetes medications, which are also often used to treat weight-loss, alleging that the drugs cause a severe stomach condition called gastroparesis, commonly known as stomach...

Ditropan Class Action Lawsuit
Ditropan (generic: oxybutynin), an overactive bladder disorder drug taken by thousands of Americans, has recently been linked to an increased risk for mental impairment and dementia, particularly among elderly patients.
Gardasil Class Action Lawsuit
Update: Gardasil Class Actions Allege HPV Vaccine Causes Autoimmune Disorders Lawyers for a large group of plaintiffs have filed a motion with the U.S. Judicial Panel on Multidistrict Litigation (JPML) requesting the creation of a new MDL Gardasil HPV vaccine...
